
Results support a “carbon memory” effect within the beach, with the evolution and migration of reaction patterns relating to the distribution of these scattered carbon pools as more mobile solutes move over them during changes in hydrologic conditions.
Kim (Adams) et al. 2019, JGR Biogeosciences
Sandy beaches are more than just recreational playgrounds for us - they’re for chemical reactions too!
It’s a mixing ground for fresh groundwater and salty seawater as waves and tides bring up seawater to the beachface.

As seawater comes up, it delivers algae, phytoplankton, and oxygen, jump starting reactions within the beach sediments.

The algae and phytoplankton — or carbon — eventually get stuck in the sand grains.
Then, as the tides move and the freshwater-seawater mixing patterns change, reactive solutes from freshwater move over the carbon pools, creating reaction patterns that are asynchronous from what the saltwater distribution tells us.
See my papers related to this effort

Spatial Patterns of Groundwater Biogeochemical Reactivity in an Intertidal Beach Aquifer
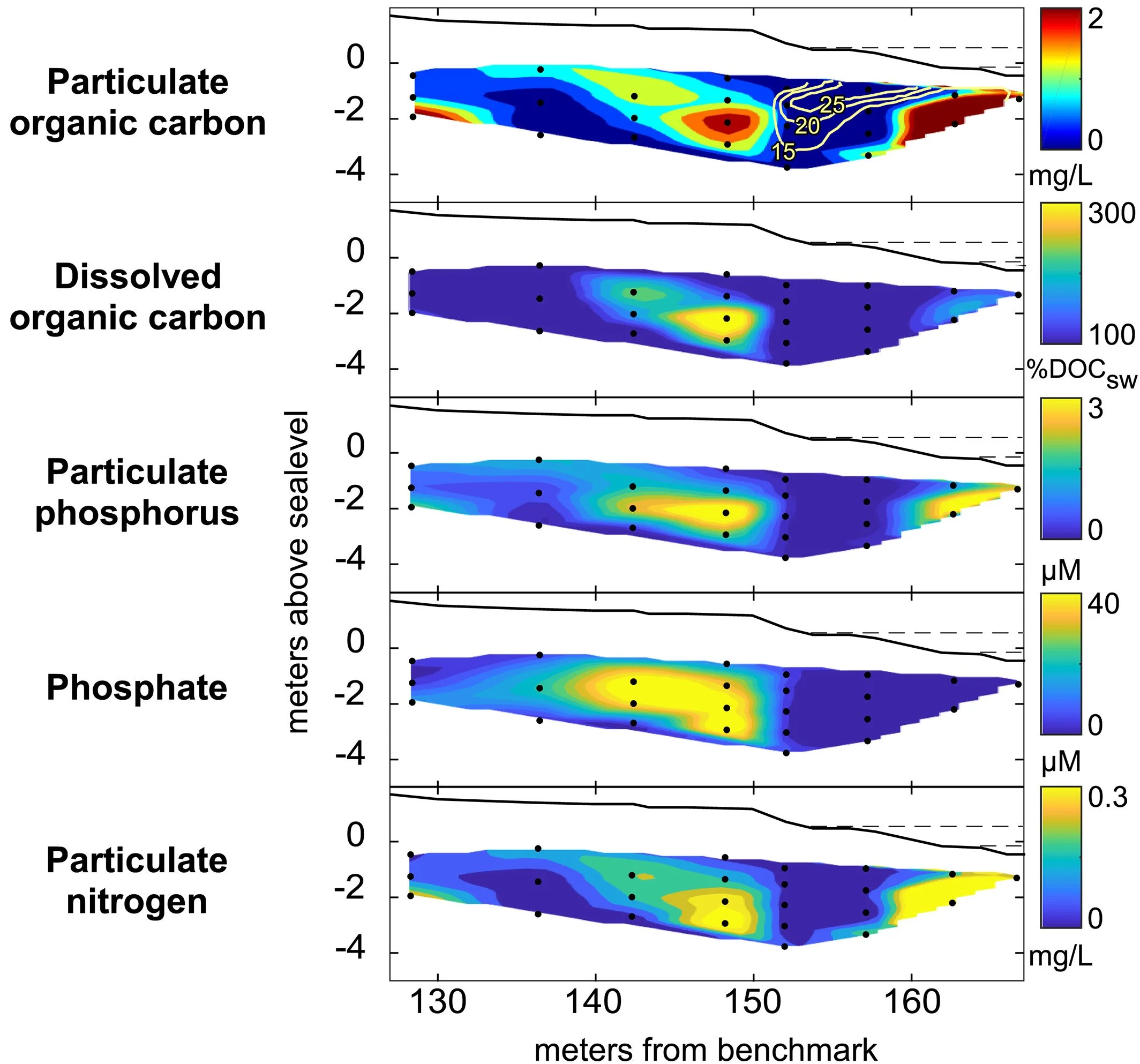
Hydrologic Shifts Create Complex Transient Distributions of Particulate Organic Carbon and Biogeochemical Responses in Beach Aquifers
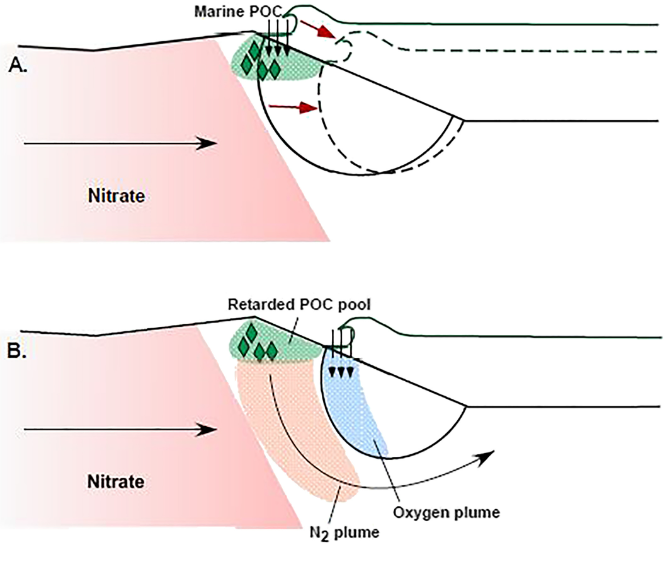
Modeling Hydrologic Controls on Particulate Organic Carbon Contributions to Beach Aquifer Biogeochemical Reactivity
